Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Nakano, Hiroko; Fuyushima, Takumi; Tsuguchi, Akira*; Nakamura, Mutsumi*; Takeuchi, Tomoaki; Takemoto, Noriyuki; Ide, Hiroshi
JAEA-Technology 2022-007, 34 Pages, 2022/06
In order to investigate the phenomenon of stress corrosion cracking (SCC) for structural materials at the light water reactor (LWR), it is important to manage a water quality for simulating high-temperature and high-pressure water. Generally, dissolved hydrogen (DH) concentration in water loop has been controlled by the bubbling method of pure hydrogen gas or standard gas with high hydrogen concentration. However, it is necessary to equip the preventing hydrogen explosion in the area installed experimental apparatus. In general, in order to prevent accident by hydrogen, it is required to take measures such as limiting the amount of leakage, eliminating hydrogen, shutting off the power supply, and suppressing combustion before an explosion occurs. Thus, the dissolved hydrogen concentration control apparatus by electrolysis method has been developed which has two electrolysis cells to control DH concentration by electrolyzing water loop. In this study, small basic experimental devices were set up. The preliminary data were acquired regarding the simple performance of two electrolysis cells and the change of DH concentration in circulation. Based on the preliminary data, the dissolved hydrogen concentration control apparatus was designed to be connected to the high-temperature and high-pressure water loop test equipment. This report describes the test results with the small basic experimental devices for the design of the dissolved hydrogen concentration control apparatus.
Ueno, Fumiyoshi
Zairyo To Kankyo, 68(1), p.2 - 8, 2019/01
It is important to control the cooling water of light water reactors (boiling water reactor and pressurized water reactor) to suitable quality in order to reduce corrosion of structural materials and generation of radioactive corrosion products. For that purpose, monitoring of water quality using electrochemical measurement method is necessary. In this article, the application of ECP measurement to BWR is mainly focused, I describe the water quality of light water reactors and the necessity of electrochemical measurement.
Takeuchi, Tomoaki; Nakano, Hiroko; Uehara, Toshiaki; Tsuchiya, Kunihiko
Nuclear Materials and Energy (Internet), 9, p.451 - 454, 2016/12
Times Cited Count:1 Percentile:10.71(Nuclear Science & Technology)no abstracts in English
Miwa, Yukio; Tsukada, Takashi
Proceedings of Symposium on Water Chemistry and Corrosion in Nuclear Power Plants in Asia 2003, p.301 - 306, 2003/00
Local composition change at grain boundaries due to radiation-induced segregation (RIS) followed by loss of corrosion resistance is considered to be a key mechanism on irradiation-assisted stress corrosion cracking (IASCC). However it is not clear that the local composition change induces the loss of corrosion resistance at grain boundaries, because RIS results in not only depletion of Cr but also enrichment of Ni and Si. This chemical composition change is different from that of thermally-sensitized stainless steels. In this study, experimental alloys were manufactured simulating the composition at grain boundaries of irradiated type 304 stainless steel and corrosion behavior of the experimental alloys was examined by weight loss measurement in 573 K water and anode polarization measurement in 1N sulfuric acid and 1mol/l sodium sulfate at 303 K. Following results were obtained: (1) In oxygenated water (DO=10ppm) at 573 K, weight loss increased with decreasing the concentration of Cr and did not depend on the concentration of Ni and Si. (2) Results of anode polarization measurements showed that alloys contained lower Cr and higher Ni and Si concentration exhibited lower corrosion potential in oxygenated, lower pH solution. In de-aerated solutions both sulfuric acid and sodium sulfate, however, there was a little influence of chemical composition on the corrosion potential.
Tanai, Kenji; Sato, Haruo; *; *
JNC TN8400 99-045, 108 Pages, 1999/11
In the anaerobic environment in the deep underground water, carbon-steel overpack corrodes and generates molecular hydrogen. It is conceivable that this hydrogen either dissolves into the porewater of the buffer and migrates through the buffer. If the rate of aqueous diffusion of hydlogen is too low compared to the rate of hydrogen generation, the concentration of hydrogen at the overpack surface will increase until a solubility limit is attained and a free hydrogen gas phase forms. It is possible that the pressure in this accumulating gas phase will increase, affecting the stability of the buffer or the surrounding rock mass. There is also a concern of possible effects on nuclide migration, as it is also conceivable that the flow of gas could push out radionuclide-bearing porewater in the buffer when it floes through the buffer. As such, experimental and analytical study must be carried out on such phenomenon to evaluate such potential phenomena. (1)Diffusion experiment of dissolved hydrogen. According to the test result concerning the effective diffusion coefficient of the dissolved hydrogen in buffer material, the effective diffusion coefficient of reference buffer material (70wt% bentonite + 30wt% sand mixture, dry density 1.6Mg m ) ranges from 10
) ranges from 10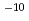 m
m s
s to 10
to 10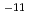 m
m s
s . The value of the effective diffusion coefficient measured for a dry density of 1.8 Mg m
. The value of the effective diffusion coefficient measured for a dry density of 1.8 Mg m is slightly smaller than the value in that for a dry density 1.6 Mg m
is slightly smaller than the value in that for a dry density 1.6 Mg m . And the effective diffusion coefficient at 60
. And the effective diffusion coefficient at 60 C tends to have slightly larger value than that at 25
C tends to have slightly larger value than that at 25 C. Test results from the foreign countries show the diffusion coefficient in the range between 10
C. Test results from the foreign countries show the diffusion coefficient in the range between 10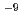 m
m s
s to 10
to 10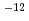 m
m s
s . Basically, these test results reported here are in the same range as these other results. (2)Gas permeability. Studies of the gas permeabinty of buffer material have been carried out by Pusch et al., Volckaert ...
. Basically, these test results reported here are in the same range as these other results. (2)Gas permeability. Studies of the gas permeabinty of buffer material have been carried out by Pusch et al., Volckaert ...
Nakano, Hiroko; Tsuguchi, Akira*; Nakamura, Mutsumi*; Tsuchiya, Kunihiko
no journal, ,
no abstracts in English
Takeuchi, Tomoaki; Nakano, Hiroko; Otsuka, Kaoru; Hirota, Noriaki; Tsuchiya, Kunihiko
no journal, ,
no abstracts in English